History of Epsom Salts
Origin
Epsom salts were discovered in Epsom, England by a farmer in 1618. He noticed his cows were not drinking the water from a certain mineral well. He tasted the water and it was bitter, he also noticed it helped the scotches and rashes on his skin. Epsom salts are made from a rock called Dolomite which is found mainly in the South Tyrollean Alps. These mountains are located in the northernmost region in Italy bordering Austria and Switzerland. The Dolomite rock belongs to a large group of substances known as "salts" creating the name Epsom salts.
Current Use:
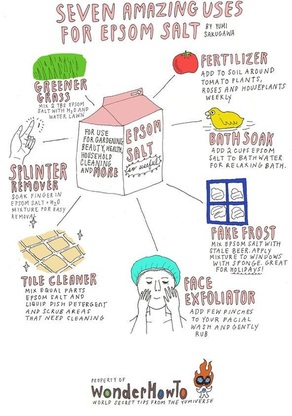
Epsom salts are very common today and can be used in a variety of different ways:
The molecular formula for epsom salt is MgSO4 this is also known as hydrated magnesium sulfate. Structurally, the magnesium is on the outside attached to the oxygen which also surrounds the central atom which is sulfur. There are two double bonds, bonding the oxygen and sulfur. Epsom salt is polar because of the uneven distribution, also all salts are polar no matter what.
Physical and chemical properties
Epsom salt crystals are transparent, odorless solid that is soluble in water. The melting point of epsom salt is 2,055 degrees (fahrenheit) and a molar mass of 120.
Toxicity
Epsom salts contain a small contain of aluminum which may be toxic which can result in kidney function problems. Also epsom salts are safe to drink because it contains one of the same minerals which is found in water this creates laxative effect by relaxing digestive muscles and also used to perform liver detox, this should be advised by a doctor.
Problems with usage or storage:
Don't use epsom salts as a laxative without the doctors permission, also pay attention to the correct amount digested because too much causes serious life-threatening side effects such as, muscle paralysis, drowsiness, difficulty breathing, etc. Overdose in taking epsom salts could result in a coma or death. Epsom salts should be kept at room temperature, no high freezing temperatures because it might effect the epsom salt. Also store in a tightly closed container stored in a cool dry are and wash after using (avoid contact with eyes).
Epsom salts were discovered in Epsom, England by a farmer in 1618. He noticed his cows were not drinking the water from a certain mineral well. He tasted the water and it was bitter, he also noticed it helped the scotches and rashes on his skin. Epsom salts are made from a rock called Dolomite which is found mainly in the South Tyrollean Alps. These mountains are located in the northernmost region in Italy bordering Austria and Switzerland. The Dolomite rock belongs to a large group of substances known as "salts" creating the name Epsom salts.
Current Use:
Epsom salts are very common today and can be used in a variety of different ways:
- used as a foot soak to soothe aches, removes odors and help get rid of rough skin
- reduces the swelling of sprains and bruises and it also helps to remove splinters
- erases stress and relaxes the body
- relieves pain and muscle cramps
- helps muscles and nerves function properly because it regulates the electrolytes in your body helping the function of nerves, enzymes and muscles
- lessens the risk of hardening arteries and blood clots because it improves blood circulation and protects the elasticity of arteries helping to prevent blood clots and reducing the risk factor of sudden deaths by heart attacks
- helps athletes foot
- exfoliates dead skin
- can be used as homemade hairspray
- fertilizes your houseplants and keeps your lawn green
The molecular formula for epsom salt is MgSO4 this is also known as hydrated magnesium sulfate. Structurally, the magnesium is on the outside attached to the oxygen which also surrounds the central atom which is sulfur. There are two double bonds, bonding the oxygen and sulfur. Epsom salt is polar because of the uneven distribution, also all salts are polar no matter what.
Physical and chemical properties
Epsom salt crystals are transparent, odorless solid that is soluble in water. The melting point of epsom salt is 2,055 degrees (fahrenheit) and a molar mass of 120.
Toxicity
Epsom salts contain a small contain of aluminum which may be toxic which can result in kidney function problems. Also epsom salts are safe to drink because it contains one of the same minerals which is found in water this creates laxative effect by relaxing digestive muscles and also used to perform liver detox, this should be advised by a doctor.
Problems with usage or storage:
Don't use epsom salts as a laxative without the doctors permission, also pay attention to the correct amount digested because too much causes serious life-threatening side effects such as, muscle paralysis, drowsiness, difficulty breathing, etc. Overdose in taking epsom salts could result in a coma or death. Epsom salts should be kept at room temperature, no high freezing temperatures because it might effect the epsom salt. Also store in a tightly closed container stored in a cool dry are and wash after using (avoid contact with eyes).
- "Ultra Epsom ™." What is Epsom Salt?. N.p., n.d. Web. 22 May 2014. <http://www.ultraepsom.com/what-is-epsom-salt.html>.
- "20 Amazing Uses for Epsom Salt." The Dr. Oz Show. N.p., n.d. Web. 25 May 2014. <http://www.doctoroz.com/slideshow/epsom-salt-uses?gallery=true>.
- "Epsom Salt Definition - Definition of Epsom Salt - What Is Epsom Salt?."About.com Chemistry. N.p., n.d. Web. 24 May 2014. <http://chemistry.about.com/od/chemistryglossary/g/Epsom-Salt-Definition.htm>.