Structure
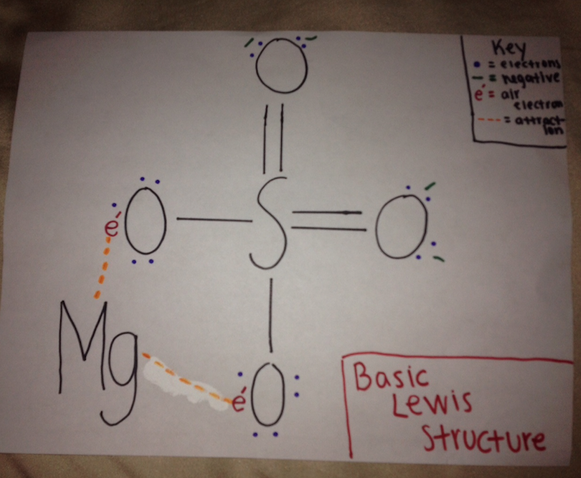
This is the basic Lewis structure showing that there is a double bond on two of the oxygen and two air electrons on the other two oxygens. The magnesium is attracted to the air electrons.
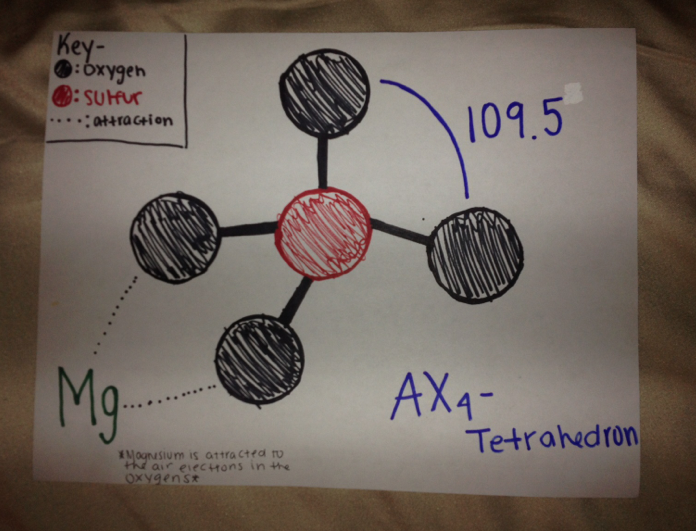
This epsom salts drawn in AX form, the SO4 in this case would be an Ax4 which is a tetrahedron creating 109.5 degree angles. The magnesium is still attracted to the oxygen in this situation. Also the molecule is polar because of the uneven distribution of electrons with the molecule.