Intermolecular Forces
Forces:
-Dipole-dipole is the strongest out of all the intermolecular forces, it only occurs in polar molecules.
-Hydrogen bonding occurs when there is fluoride, nitrogen and nitrogen are involved in bonding because of electronegativity.
-Dispersion is the weakest force and occurs when the electrons in two adjacent atoms involve positions that make the atoms form temporary dipoles, this is commonly found in non polar molecules.
-Dipole-dipole is the strongest out of all the intermolecular forces, it only occurs in polar molecules.
-Hydrogen bonding occurs when there is fluoride, nitrogen and nitrogen are involved in bonding because of electronegativity.
-Dispersion is the weakest force and occurs when the electrons in two adjacent atoms involve positions that make the atoms form temporary dipoles, this is commonly found in non polar molecules.
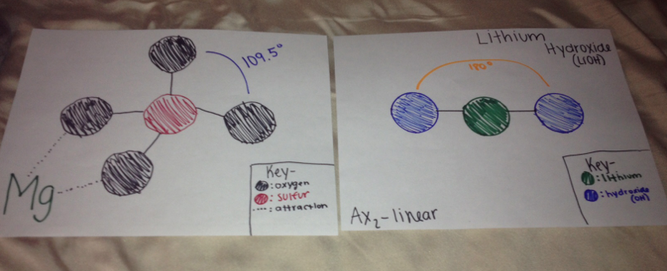
If magnesium sulfate was put to react with lithium hydroxide two forces would be acting in this reaction. First, dipole-dipole, this force occurs in all polar molecules since both molecules are polar the dipole-dipole force is acting upon this reaction. Also hydrogen bonding because the hydrogen in lithium hydroxide would bond or be attracted to the oxygen in the magnesium sulfate.